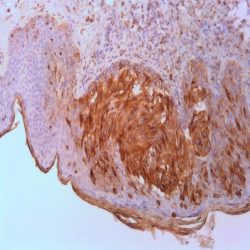
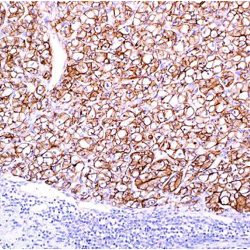
Name:Antibody S100-Beta Protein clone SP127
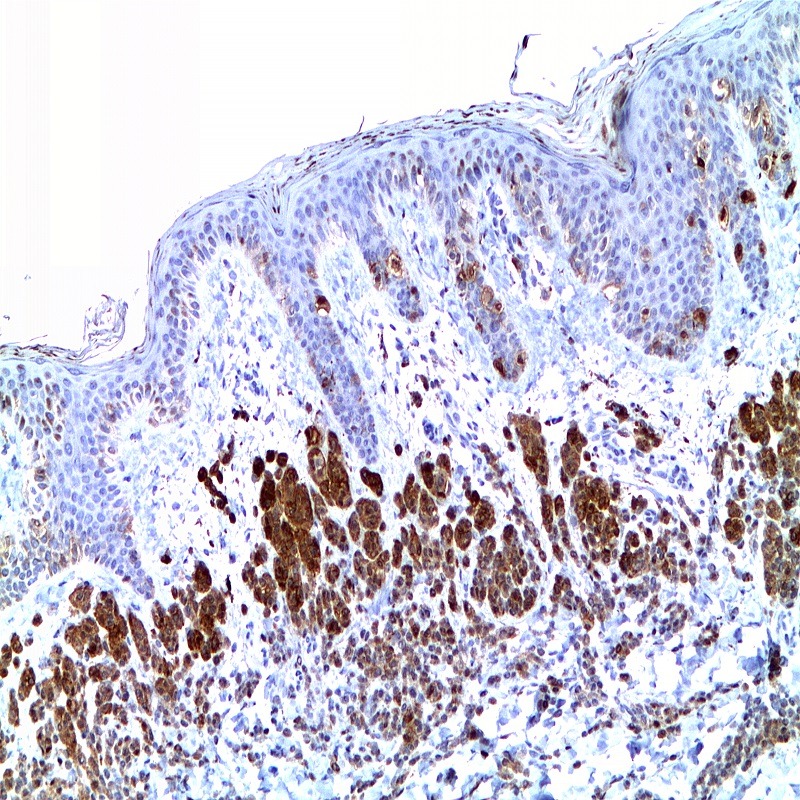
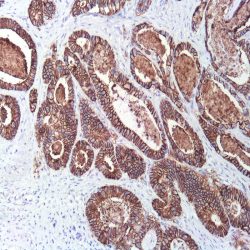
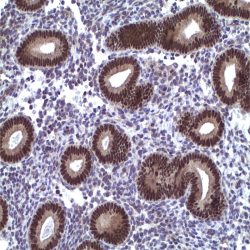
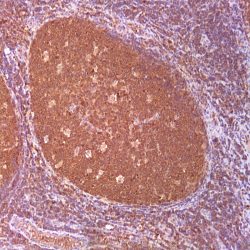
Description and applications: S100 proteins are a set of 25 small acidic molecules with a molecular mass of 10-12 kDa that are functionally grouped into homo or heterodimers composed of two subunits, alpha and beta, capable of combining with each other and with extensive sequence homology. There exist up to 14 variants of the alpha chain gene, which is located on chromosome 1q21; on the contrary, there is only one sequence of the beta chain gene, located on chromosome 21q22.3, showing its greater functional specificity. In its dimeric form, the S100 protein, together with calmodulin and troponin C, belongs to the calcium binding protein family; its affinity for this ion, as well as for other metals such as zinc, is remarkable. It is for this reason that the S100 protein is involved in basic cellular activities such as cation diffusion across lipid membranes, microtubule assembly, and RNA polymerase activity. In neurons, the S100 protein also regulates the interaction between chromosomes and synaptosomes. Depending on the combination between the alpha and beta single chains to form dimers and the homology of their sequences, there exist three forms of S-100 protein: A, with at least 15 subtypes between A1 and A14, B, and G. The S100-B protein is a 21-kDa beta-chain homodimer involved in the regulation of numerous processes that require calcium ion flow stimulation. The S100-B protein is present in neurons, glial cells of the central nervous system, Schwann cells and satellite cells of the peripheral nervous system (but not perineural cells), melanocytes, myoepithelial cells, glandular epithelia of the breast and the kidney, skeletal muscle and heart cells, adipocytes, and chondrocytes. This molecule is also expressed in antigen-presenting cells such as Langerhans cells of the skin, and interdigitating dendritic cells in the T-cell areas of lymphoid tissue. In contrast, the S100-B protein is not present in smooth muscle fibres. In neoplasms, the S100-B protein is highly expressed in tumours of the central and peripheral nervous systems and in melanomas, including desmoplastic melanoma. However, up to 4% of melanomas may be negative against this antibody, suggesting the need to assess this staining within a minimal panel of antimelanoma antibodies that at least includes the HMB45 and Melan A markers. Likewise and in comparison with primary lesions, some loss of expression of the S100-B protein has been observed in metastatic melanoma lesions. The S100-B protein is also useful in the diagnosis of tumours derived from cartilage, adipose tissue, and interdigitating reticular cells in lymphoid tissue, as well as granular cell peripheral nerve sheath tumours, myoepithelial tumours, paragangliomas (sustentacular cells), PEComas, Rosai-Dorfman disease, clear cell sarcoma, chordoma, adenoid cystic carcinoma, and isolated cases of synovial sarcoma and rhabdomyosarcoma. Atypical fibroxanthomas, cardiac sarcomas, myofibroblastomas, xanthomas, and solitary fibrous tumours are all negative for S100-B. Finally, altered overexpression of the beta-chain gene with S100-B protein overexpression may be found in the basis of some of the lesions observed in various types of neurological and genetic diseases such as Alzheimer’s disease, Down’s syndrome (trisomy 21), epilepsy, amyotrophic lateral sclerosis, and type I diabetes, in which an excess of the S100-B protein has a potential neurotoxic effect. In this sense, several tests have been developed for serum determination of this cell damage marker in these patients.
Composition: Anti-human S100-Beta Protein rabbit monoclonal antibody purified from serum and prepared in 10mM PBS, pH 7.4, with 0.2% BSA and 0.09% sodium azide

دیدگاهها
هیچ دیدگاهی برای این محصول نوشته نشده است.