Name: CD45RA Monoclonal Antibody clone SPM504
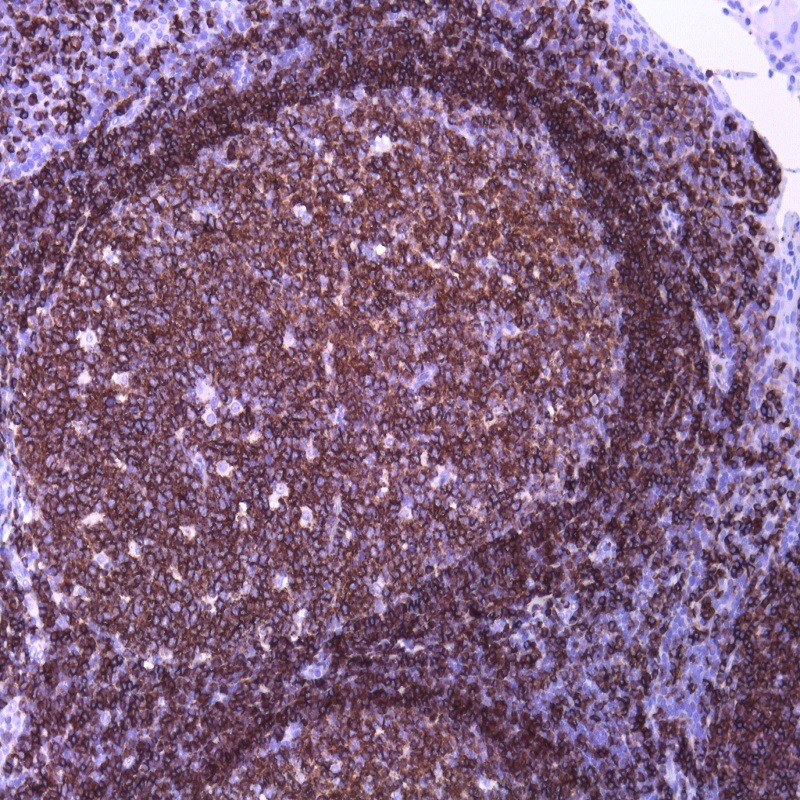
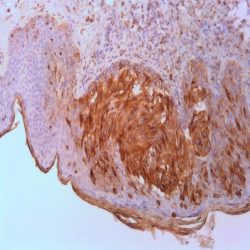
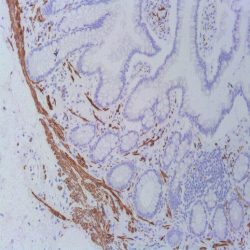
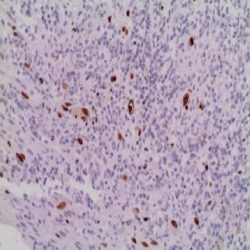
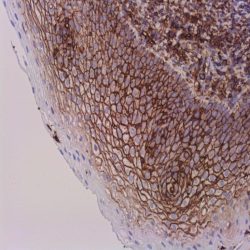
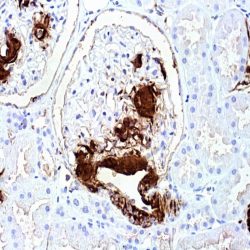
Description and aplications: This antibody reacts with the CD45RA isoform with a molecular mass of 220 kD. CD45RA is present in peripheral blood B cells, follicular mantle and centre, medullary thymocytes, monocytes, and mature T cells that have not been modulated by antigenic contact. In this regard, and after immune processing in the thymus, T cells are released into peripheral blood to colonize secondary lymphoid organs, including spleen, lymph nodes, and mucosa-associated lymphoid tissue (MALT). These lymphocytes, not yet activated in vivo by antigens (naive cells), are phenotypically characterized by the co-expression of the highest molecular mass CD45 isoforms, CD45RAC and CD45RA, and CD62L (peripheral lymph node homing receptor), a decisive molecule for the fixation of cells in thymus-dependent areas of secondary lymphoid organs. When these cells are stimulated by its specific antigen expressed by the corresponding antigen-presenting cells and are transformed into effector T cells, either CD4- or CD8-positive or memory T cells, CD45RA and CD62L expression disappears, the former being replaced by the lower molecular mass CD45R0 isoform, and the latter, by various type 1 and type 2 integrins and APO-1/Fas receptor (CD95), an apoptosis inducer. The CD45 molecule, also called leukocyte common antigen, is a membrane glycoprotein that occupies up to 10% of the surface of cells expressing it. In mammals, its homology varies markedly between the intracytoplasmic region (greater than 90%) and the extracellular region (only 35%, although with similar domain organization). Structurally, there are multiple CD45 isoforms determined by the different possible ways of joining exons 4, 5, and 6 during the synthesis of the mRNA encoding the extracellular domain of the protein. These isoforms are referred to as A, B, and C, using the RABC terminology for the bigger isoform, which includes all three exons, RA and RB when the exonic expression is restricted to one of these exons, and RO to designate the smaller molecule, which lacks all three exons. The sequence of these three exons includes multiple oxygen-linked glycosylation sites that can be variably modified by sialic acid. This, on the one hand, causes the molecular mass of the different isoforms to vary between 180 kD (RO) and 240 kD (RABC); on the other hand, it gives these molecules remarkable differences in terms of their shape and anionic charge. The rest of CD45’s extracellular domain shows marked nitrogen-linked glycosylation and contains a cysteine-rich region followed by three amino acid repeats analogous to fibronectin type III domain. CD45’s high degree of glycosylation, attributable to both sialic acid and oligosaccharides on the variable domains and N-glycoconjugates on the constant extracellular domain, plays an important role in the functional activity of the molecule, as demonstrated by the interaction between CD45RO of T cells and B cells expressing CD22 or the mannose hybrids on CD45 in the development of immature thymocytes. The rest of the CD45 molecule consists of a single transmembrane domain followed by a long intracytoplasmic tail containing two tandem-placed identical domains (D1 and D2) with protein-tyrosine phosphatase (PTPase) homology. Of these domains, only D1 possesses enzymatic activity capable of rescuing TCR signal activation in CD45-deficient cell lines. The function of D2 is hardly understood, although it seems to contribute to the molecule’s intracytoplasmic stability. Although in in vitro models the extracellular domain of CD45 is not essential for its intracytoplasmic function, the exonic transcription of this domain is strongly regulated depending on the cell type that expresses it, its development, and its degree of activation, thus playing an important functional role in vivo.The CD45RA epitope recognized by the MB1 antibody is useful for the study of B-cell lymphomas; however, as some of them are not positive against it, it is advisable to use it as part of a large panel of antibodies. Because the epitope recognized by MB1 may be altered by prolonged formol fixation, and in order to ensure a correct interpretation of results, it is appropriate to check that at least some normal B cells in the histological section are stained. The antibody shows no reactivity against cortical thymocytes, immature T cells, and activated B cells. This antibody reacts against human tissue. In other species, it has not been tested.
Composition: Anti-human CD45RA mouse monoclonal antibody purified from serum and prepared in 10mM PBS, pH 7.4, with 0.2% BSA and 0.09% sodium azide

Reviews
There are no reviews yet.